Pests & Diseases
These are the major pests and diseases that affect maize crops in New Zealand
FILTER:
EFFECT
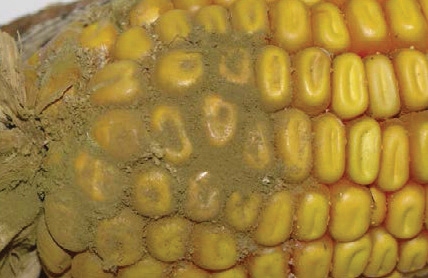
Symptoms: Olive-green coloured powdery mould on the base or tip of infected ears.
Toxin type: Aflatoxin
Common smut

EFFECT
Symptoms: Galls form on ears, leaves, stalks and/or tassels of infected plants.
Toxin type: N/A
Diplodia ear rot

EFFECT
Symptoms: Grey to greyish-brown mould often starting at the base of the ear spreading to cover much of the infected ears. Infected ears appear mummified and weight is severely reduced.
Toxin type: N/A
Fusarium ear rot

EFFECT
Symptoms: White, grey or pink coloured fungal growth on individual kernels or patches of kernels in infected ears.
Toxin type: Fumonisin
Fusarium Head Blight

EFFECT
Caused by a number of Fusarium species, with the disease occurring on crop residues and spreading to the growing plant. Symptoms appear after flowering and infects the cob via the silks, forming a mycelium sheath starting at the apex of the ear and progressing downwards. The decomposing kernels and the mycelium produce a characteristic pinky-brown colouring. Often an attack can occur opportunistically, following corn earworm damage or similar. There is potentially some varietal resistance, but management of previous crop residue is the key to control.
Gibberella ear rot

EFFECT
Symptoms: White to salmon/pink mould starting at the tips of ears, with husks tightly adhering to infected ears.
Toxin type: Deoxynivalenol and Zearalenone
Head Smut

EFFECT
Head smut can cause serious yield losses. The cobs and tassels are invaded by spores, taking over the zone of the kernels and stamens. It is more prevalent in low-lying valleys or on land prone to flooding and in sandy or silty soils. Affected cobs are bulbous, soft and with no visible silks. The fungus gets into the plant through the mesocotyl and roots before the 8-leaf stage and spreads systemically, going up to the developing cob and tassel. Cold temperatures will slow the rate of germination and establishment of the pathogen, which survives in heat. Disease development is helped by any factor that slows early growth, for example, early dry conditions or compacted soil.
Protection is in the form of hybrid resistance and fungicidal seed treatment. Seed crops are monitored carefully to ensure no carry-over of infection.
Penicillium ear rot

EFFECT
Symptoms: Blue-green powdery mould on and between the kernels, usually near the tip of infected ears.
Toxin type: N/A
Trichoderma ear rot

EFFECT
Symptoms: Dark-green fuzzy mould grows on and between the kernels of infected ears.
Toxin type: N/A
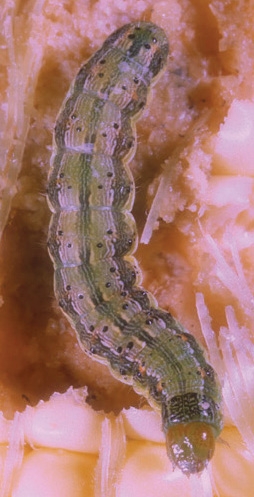
Fall armyworm (FAW)

EFFECT
The first signs of a FAW infestation are usually identified by small pinhole-like feeding marks that look like windows anywhere on the leaf but seem to be prominent around the leaf collars. As the larvae grow, the hole size increases and to a depth that will break through to the other side of the leaf.
Severe infestation can cause severe crop defoliation. FAW larvae attack the ear, silks and kernels which will have detrimental effects on cob development and leaf mass, ultimately reducing crop yield.
While FAW can be difficult to distinguish from the other Army Worms present in NZ maize crops (Cosmopolitan Army Worm, Tropical Army Worm), mature FAW larvae have several distinct features:
- A dark head with an upside down, pale Y-shaped marking
- Four raised spots shaped like a trapezium on each body segment (seen from above)
- Four sports forming a square on the second-to-last body segment
EFFECT
Raised, rust coloured pustules appear on both sides of the leaf blade.
Chlorosis and leaf death may occur in severe infections on susceptible hybrids.
The disease favours humid conditions and moderate temperatures (15 to 25°C), including dewy nights/mornings and overwinters on host oxalis species.
EFFECT
Eyespot appears as a large number of small circular spots, usually less than 4 mm in diameter and often just confined to the leaves.
Eyespot is cream to grey coloured in the centre, with a purply -brown edge and surrounded by a translucent halo when seen against the light. Initial symptoms can appear at the seedling stage and spread to the lower leaves; but the disease does not become fully established until after flowering, when it attacks the leaves above the cob. Leaf loss is the result, with severe infection causing kernels to shrivel.
Lower summer temperatures and leaf surface moisture help the disease to manifest itself, with several hours of leaf moisture required for the disease to spread. The fungus survives easily on crop residues, so it is important to bury the trash if the disease is present in a mono-crop situation.
Northern Leaf Blight

EFFECT
Leaf blight is an endemic leaf disease, with visible symptoms in the form of large spindle-shaped lesions, running in the direction of the veins and causing premature drying of the leaves. In damp weather, dark brown fructifications develop, causing the disease to spread, with airborne spores infecting the upper leaves. The impact on the crop occurs through the loss of leaf area for photosynthesis and can happen rapidly – 5-12 days from initial infection to visible symptoms.
NLB is often restricted to certain specific regions of New Zealand due to the occurrence of the required climatic conditions. Risk is increased with the following factors: planting in a known hotspot, planting in a paddock subject to a previous outbreak or in close proximity to one, and close to a grain dumping site or a grain storage facility.
Control is helped by hybrid selection and shredding/mulching of previous crop residues.
Argentine Stem Weevil

EFFECT
Can be a major threat to seedling maize plants.
Larvae enter the maize shoot soon after emergence then bore towards the base of the plant and sever the growing point.
ASW feeds on pasture tillers, therefore maize crops sown into paddocks previously in pasture or with pasture weeds with a short cultivation period, are most at risk.
Visible damage includes emergence failure, wilting and collapse.
Present in all New Zealand maize growing regions.
Damage is caused by the larvae, small white legless grubs (1 to 3 mm long) which transfer from decaying grass tillers before or soon after maize crop emergence.
Damage includes emergence failure, wilting and death of plants up to V4.
Death of emerged seedlings is characterised by a small hole of the base of the stem and subsequent wilting of the whorl leaves followed by either plant death or prolific tillering.
Imidicloprid (Gaucho) provides some level of control and Clothianidin (Poncho®) usually provides an adequate control of ASW larvae, but in severe infestations significant damage may still occur.
A grass free fallow period of 2 to 3 weeks should significantly reduce the infestation load.
Black Beetle

EFFECT
Confined to the warmer (upper) regions of the North Island. Infestations are more severe following long term pasture, particularly paspalum or kikuyu.
Damage is characterised by signs of chewing of the roots and stem at the base of plant. Damaged plants typically show signs of wilting and either die or tiller from the base of the plant and become stunted.
Both Imidicloprid (Gaucho) and Clothianidin (Poncho®) seed treatments provide adequate levels of control of black beetle.
EFFECT
Also known as the tomato fruitworm can cause significant damage to developing cobs by feeding on silks (during pollination) and kernels throughout the grain fill period (R1 to R5) and subsequently by fungal attack of the grain during drydown (R6 onwards). Caterpillars are variable in colour ranging from pink to green or brown, with dark, bristley, raised bumps.
Controlling corn earworm is difficult once the caterpillars have penetrated the ear, but prior to this they may be effectively controlled by application of a synthetic pyrethroid spray whilst being mindful that this will also reduce the numbers of beneficial insects present in the crop; this includes the parasitic wasps Apanteles kazak (moderately effective at controlling corn earworm) and Apanteles rufricus (controls cosmopolitan armyworm).
EFFECT
Caterpillars are usually found at or shortly before silking. CAW caterpillars graze on leaf laminae of maize and leave only the midribs in severe cases.
CAW will consume the bottom half of plants before moving to the upper parts.
Silks and seeds may also be consumed.
Greasy Cutworm

EFFECT
Cutworm damage is determined by the number and growth stage of the caterpillars in relation to the crop growth stage.
The first stages of a cutworm's life is spent on the soil surface feeding on plant leaves. As the cutworm grows it begins to sever plants and consume them, this is referred to as the “cutting stage”.
The most devastating effect occurs where “cutting stage” coincides with crop emergence. Damage though is more frequently caused by caterpillars tunnelling up the maize stem after the 4-leaf stage.
EFFECT
Found throughout the North Island and warmer parts of the South Island the green vegetable bug (GVB) overwinters as adults and lay eggs in early spring.
There are typically two or more generations per season and although they are not usually a major concern from a yield perspective in maize, they do cause damage when feeding on kernels by causing discolouration, shrinkage and potential entry points for infection by fungi associated with kernel rots.
There are 3 parasitoids (all wasps) in New Zealand that are known to infect GVB, as well as a few predators (e.g. starlings and german wasps).
There are no chemicals registered for use on GVB in maize.
Slugs

EFFECT
Slugs may damage emerging maize crops. Cavities are gouged into the seed, sometimes affecting the growing point, and early leaves often appear shredded as a result of slug feeding. Crops sown on heavy, moist soils which have a cloddy structure and direct drilled crops are most at risk.
Wireworm

EFFECT
Wireworms are long (up to 30 mm), segmented, six legged insect larvae with yellowish to brown colouring. Several species of wireworm exist in NZ pastures.
Damage to maize is similar to that caused by ASW larvae, in that they tunnel into the base of the plant causing the centre leaves to wilt and die.
Currently there is no registered chemical to control wireworm in New Zealand. Intensive cultivation (disc and power harrow) may also help to reduce the infestation load by physically damaging the insect larvae and exposing them to predators (e.g. birds).
Root Necrosis

EFFECT
Rhizoctonia/Fusarium - Root rot diseases are caused by a number of different pathogenic fungi affecting the roots. Rhizoctonia affects main and brace roots, causing severe necrosis with a blackening of the affected areas. Fusarium fungi causes rotting with a characteristic browning of the roots. Visible effects in the crop are often seen during early grain fill with leaves turning to green-grey, then to pink before drying out. The plant is weak, easy to pull out and therefore prone to lodging.
Higher risk situations are: sandy soils, irrigated land and in single-crop conditions where there are soil structure problems.
EFFECT
Shiny, black discoloration that cannot be scratched off the rind.
Bacterial stalk rot

EFFECT
Dark brown water soaked lesions at base of stem with soft, slim stalk tissues accompanied by a strong odour.
Charcoal rot

EFFECT
Tiny, black sclerotia produced inside the stalk gives the appearance of charcoal dust.
Diplodia stalk rot

EFFECT
Tiny, raised black dots (pycnidia) on lower nodes are embedded in the stalk and cannot be scraped off.
Fusarium stalk rot

EFFECT
Pinkish-white fungal growth on outside of stalk, pink or salmon coloured discolouration on inside of stalk, lack of visible reproductive structures, crowns often are brown and rotted.
Gibberella stalk rot
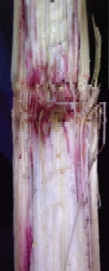
EFFECT
Bright pink to red discolouration at the nodes.